UPDATES
IBS Center for Multidimensional Carbon Materials
Total : 62, Now : 6 page
전기차 5배 빠르게 충전할 수 있는 산호 닮은 신소재 나왔다
고속 충전‧대용량 가능한 리튬이온배터리 음극 소재 개발
'바다의 꽃'으로도 불리는 산호를 꼭 닮은 신소재가 개발됐다. 기초과학연구원(IBS) 다차원 탄소재료 연구단은 박수진 포항공대 교수팀과의 공동연구를 통해 리튬이온배터리 충전 속도를 5배 높일 수 있는 새로운 음극 소재 물질을 개발했다.
▲ IBS 다차원 탄소재료 연구단 산호와 모습이 꼭 닮은 신소재를 개발했다. (출처: PEXELS)
전기자동차 등 고성능을 요구하는 제품이 늘어나며 더 빠르게 충전되면서도 대용량인 배터리 개발이 요구되는 상황이다. 하지만 현재 리튬이온배터리의 음극에 사용되는 소재인 흑연은 이론적 용량 한계가 있을뿐더러, 고속충전 시 음극 표면에 리튬 금속이 석출돼 배터리 전체의 성능을 낮춘다는 문제가 있다.
과학자들은 흑연을 대체할 소재로 실리콘에 주목했다. 실리콘은 흑연보다 용량이 10배 이상 커 고에너지 배터리로 적합하기 때문이다. 아직 남은 숙제는 충‧방전 시 부피 변화가 커서 잘 깨지는 실리콘의 고질적 문제를 해결하는 것이다.
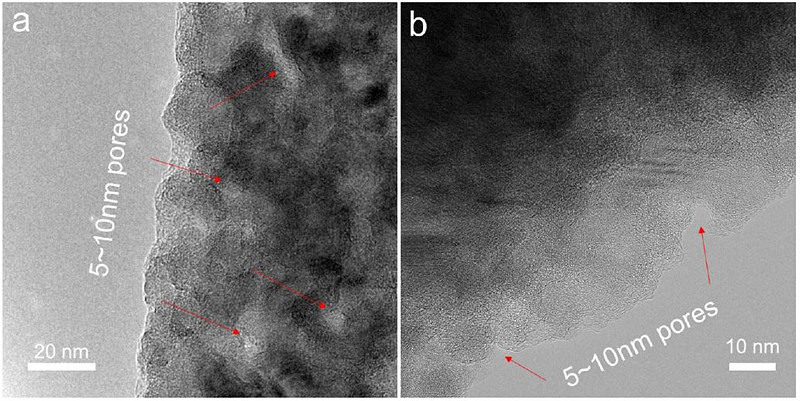
▲ 연구진이 새로 개발한 전극의 구조. 다공성 실리콘 나노와이어를 산호 모양처럼 이어 붙여 만든다. (출처: ACS Nano)
공동연구진은 물질 단계부터 새로운 설계를 제안하며 이 문제를 해결했다. 우선 구멍이 많은 실리콘 나노와이어 구조체를 재료로 사용해 실리콘의 부피 팽창 문제를 완화했다. 내부 구멍들은 충전 시 팽창한 실리콘을 받아들여 실리콘이 깨지지 않고 견디도록 돕는다.
이후 다공성 실리콘 나노와이어를 높은 밀도로 연결시키고, 여기에 탄소를 나노미터 두께로 얇게 씌웠다. 이렇게 만들어진 산호 모양의 '실리콘-탄소 복합체 일체형 전극'은 전기전도도가 향상돼 고속 충전이 가능했다.
이번 연구의 공동 제1저자인 빈 왕 IBS 다차원 탄소재료 연구단 연구위원은 "산호 모양 실리콘의 다공성 구조는 배터리에 적용했을 때 리튬이온을 빠르게 전달하도록 돕고…
작성자 : CMCM
2019.03.26

고품질 흑연, 그래핀 따라 도미노처럼 줄맞춰 만든다
- IBS 다차원 탄소재료 연구단, 단결정 흑연 개발 초석 다져 -
연필심의 재료로 친숙한 흑연은 철강, 반도체부터 태양광, 에너지 저장 등 여러 산업에 널리 쓰이는 원료다. 전기적 특성이 우수하고, 고온에도 잘 견디는 것은 물론, 가격까지 저렴하기 때문이다. 물성이 우수한 흑연을 사용할수록 제품의 성능을 높이기도 유리하다.
기초과학연구원(IBS) 로드니 루오프 다차원 탄소재료 연구단장(UNIST 특훈교수) 팀은 단결정 그래핀을 이용해 고품질 흑연을 제조할 수 있는 새로운 기술을 개발했다.
▲단결정 그래핀(붉은 상자) 주변에 결정 방향이 비슷한 고품질 흑연이 만들어지는 장면
자연에서 발견되거나 인공적으로 합성한 흑연은 대부분 다결정 구조를 가진다. 재료를 이루는 결정 속 원자의 배열이 일정하지 않고 제각각이라는 뜻이다. 이런 다결정 구조는 결정들이 만나는 경계면에 결함이 존재해 물질 고유의 특성이 저하된다는 단점이 있지만, 아직까지 대면적 단결정 흑연을 만드는 기술은 개발되지 않았다.
다차원 탄소재료 연구단 연구진은 단결정 그래핀을 흑연의 중심에 두고, 높은 온도를 가하는 방식으로 고품질 흑연을 제작했다. 이때 흑연은 단결정 그래핀에 가까운 영역에서부터 고배향성을 가진 고품질 흑연으로 재탄생했다. 무질서하게 배열돼 있던 탄소원자들이 그래핀의 구조를 복사해 도미노처럼 재배열했다는 의미다.
▲고온으로 열처리하는 과정의 초기에 그래핀 층 주변으로 고배향성 흑연이 생기는 장면
대면적 단결정 흑연의 합성은 학계에서 도전적인 과제로 꼽힌다. 이번 연구의 공동 제1저자인 빈 왕 IBS 다차원 탄소재료 연구단 연구위원은 "고품질 흑연 제작은 전적으로 단결정 그래핀의 크기에 달렸다"며 "연구진은 현재 가로 4cm, 세로 8cm의 단결정 그래핀을 제작하는데 성공했고, 이후 더 대면적화할 계획"이라고 말했다. 향후 더 큰 고배향성 흑연 필름을 제작할 수 있다는 의미다.
…
작성자 : CMCM
2019.03.26

Novel Technology for Highly Ordered Arrays of 'Graphene Quantum Dot'
Their findings have been published in the January 2019 edition of Nature Communications.
A new study, affiliated with UNIST has introduced a novel technology, capable of fabricating highly ordered arrays of graphene quantum dot (GQD). The new technology is expected to pave the way for many other types of devices and physical phenomena to be studied.
This breakthrough has been led by Professor Hyeon Suk Shin in the School of Natural Science at UNIST. In their study, the research team demonstrated a novel way of synthesizing GQDs, embedded inside the hexagonal boron nitride (hBN) matrix. Thus, they demonstrated simultaneous use of in-plane and van der Waals heterostructures to build vertical single electron tunnelling transistors. Their findings have been published in the online edition of Nature Communications on January 16, 2018.
Graphene quantum dots (GQDs) have received much research attention, due to their un…
작성자 : CMCM
2019.01.24
![[IBS People] 상상 이상의 탄소를 연구하는 과학자](/data/file/notice/1917193931_GX1OFRLy_ca1e7274d0c9f2e05e7ade116cf3cce09c0410ae.png)
상상 이상의 탄소를 연구하는 과학자
- 펑딩 IBS 다차원 탄소재료 연구단 그룹리더 -
"Welcome to the Feng Ding Kingdom :-)"
울산 UNIST에 위치한 기초과학연구원(IBS) 다차원 탄소재료 연구단의 한 연구실. 탄소 구조물을 이리저리 만지작거리던 펑딩 그룹리더(UNIST 교수)가 이내 장난스러운 표정으로 왕관 모양을 만들어냈다. 소년 같은 미소를 자랑하는 펑딩 교수는 탄소 이론 및 컴퓨터 시뮬레이션 분야 석학이다. 세계 곳곳에서 출간된 탄소를 소재로 한 연구논문엔 그의 이름을 심상치 않게 발견할 수 있다. 탄소와 함께한 지 20여 년. 그가 '과학자들의 유명인사'로 자리매김한 이유는 뭘까.
한국에 정착한 모태 기초과학자
"과학자라는 꿈을 꾸게 만든 특별한 계기조차 없었던 것 같아요. 아마 기초과학자가 되기 위해 태어난 것은 아닐까요? 모태 과학자라고나 할까."
[피플 인터뷰 영상보기] 펑딩 IBS 다차원 탄소재료 연구단 그룹리더
어릴 때부터 수려한 외모를 자랑하며 떡잎부터 다른 자태를 뽐낸 사람을 '모태 미녀'라고 한다. 연예계에 모태 미녀가 있다면 IBS에는 모태 과학자가 있다. 펑딩 교수는 학창시절부터 줄곧 과학교과목 성적이 좋았다고 한다. 교과서 속 과학지식이 자연에서 벌어지는 모든 사건을 설명한다는 점이 그의 흥미를 끌었다. 과학이론을 공부하고 연구하게 된 것은 그에겐 그저 자연스러운 일이었다.
IBS 다차원 탄소재료 연구단에 합류하기 전, 펑딩 교수는 홍콩폴리텍대의 영년직 교수로 일했다. 명성 높은 학교, 좋은 학생들과 함께하는 연구는 꽤 완벽한 삶처럼 느껴졌다. 하지만 한 가지 열망이 있었다. 기초과학 연구에만 온전히 몰입하고 싶다는 열망이었다. 로드니 루오프 IBS 다차원 탄소재료 연구단장의 전화 한통에 그의 한국행은 부지불식간에 결정됐다. 두 사람은 학회에서 안면을 익힌 적은 있었으나, 개인적인 친분이나 교류는 없었다.
"기초과…
작성자 : CMCM
2018.12.28

IBS Announced "IBS Declarations for Another Great Leap Forward"
▲ The Scientific Advisory Board (SAB) members, IBS directors, and the IBS president had in-depth discussion on the roles, responsibilities, and future directions for IBS to move forward.
The Institute for Basic Science (IBS) held 'IBS Forum for Another Leap Forward' on October 12, 2018. The Scientific Advisory Board (SAB) members, IBS directors, and the IBS president had in-depth discussion on the roles, responsibilities, and future directions for IBS to move forward.
The declarations pronounce IBS commitments and hopes for IBS to make another great leap forward to become a global research institution.
IBS commitments include its efforts i) to serve as a catalyst for research collaboration at home and abroad; ii) to ensure scientific excellence as the value that we cannot compromise on; iii) to foster and bring in young talents; iv) and to communicate with the civil society to earn their s…
작성자 : CMCM
2018.10.23

Producing Defectless Metal Crystal of Unprecedented Size
- IBS scientists report in Science a new method to obtain large single crystals up to 32 ㎠, made of copper, nickel, cobalt, platinum, or palladium -
A research group at the Center for Multidimensional Carbon Materials, within the Institute for Basic Science (IBS), have published in Science about a new method to convert inexpensive polycrystalline metal foils to single crystals with superior properties. It is expected that these materials will find many uses in science and technology.The structure of most metal materials can be thought of as a patchwork of different tiny crystals, bearing some defects on the borders between each patch. These defects, known as grain boundaries (GBs), worsen the electrical and sometimes mechanical properties of the metal. Single crystal metals, instead, have no GBs and show higher electrical conductivity and other enhanced qualities that can play a major role in multiple fields, such as electro…
작성자 : CMCM
2018.10.22
